|
Chemical analysis of the formation shows 10 wt% Fe, primarily in the +2 valence. The reservoir has an estimated storage capacity of 100 giga tonnes of carbon dioxide and comprises layered basalt flows with sublayering that generally alternates between low permeability massive and high permeability breccia. Recently this nonpotable and extensively characterized basalt formation is being reconsidered as a potential reservoir for geologic sequestration of carbon dioxide. For several decades the United States Department of Energy has been investigating Columbia River basalt in the Pacific Northwest as part of its environmental programs and options for natural gas storage. In addition to being globally abundant and vast, deep basaltic lava formations offer mineralization kinetics that make more » geochemical trapping a dominate mechanism for trapping carbon dioxide in these formations.  
In sedimentary saline formations the dominant mechanisms are structural and dissolution trapping, with moderate to weak contributions from hydrological and geochemical trapping where, hydrological trapping occurs during the imbibition of aqueous solution into pore spaces occupied by gaseous carbon dioxide, and geochemical trapping is controlled by generally slow reaction kinetics. The principal mechanisms for the geologic sequestration of carbon dioxide in deep saline formations include geological structural trapping, hydrological entrapment of nonwetting fluids, aqueous phase dissolution and ionization, and geochemical sorption and mineralization. 0000546930 - ZLNQQNXFFQJAID-UHFFFAOYSA-L - Magnesium carbonate USAN - Similar structures search, synonyms, formulas, resource links, and other chemical information. The most difficult challenge is to overcome the low permeability of typical deep-sea sediments, and a variety of approaches are suggested for future research. Our analysis has shown the feasibility of this type of storage, and also emphasizes that escape or leakage from such sites would be negligible. Over the course of the three-year award, our results support an important role for sub-seabed storage in a diverse portfolio of carbons sequestration options. In addition, we are investigating the kinetics of calcium carbonate dissolution in the presence of CO storage in deep sea sediments using a range of approaches including experiments, permeability analysis, and modeling. Our modeling efforts in the first year show that the idea is feasible, but requires more sophisticated analysis of fluid flow at high pressure in deep sea sediments. Through laboratory and modeling efforts, we are studying the flow of liquid carbon dioxide and carbon dioxide-water mixtures through calcium carbonate sediments to better understand the geomechanical and structural stability of the sediments during and after injection. Our goal is to evaluate the basic feasibility of this approach, including an assessment of optimal depths, sediment types, and other issues related to site selection. The NIST Chemistry WebBook (see link below) is an online resource that contains standard enthalpy of formation for various compounds along with the standard absolute entropy for these compounds from which the standard Gibbs free energy of formation can be calculated.Our research is aimed at investigating several technical issues associated with carbon dioxide sequestration in calcium carbonate sediments below the sea floor through laboratory experiments and chemical transport modeling. 
Far more extensive tables can be found in the CRC Handbook of Chemistry and Physics and the NIST JANAF tables. 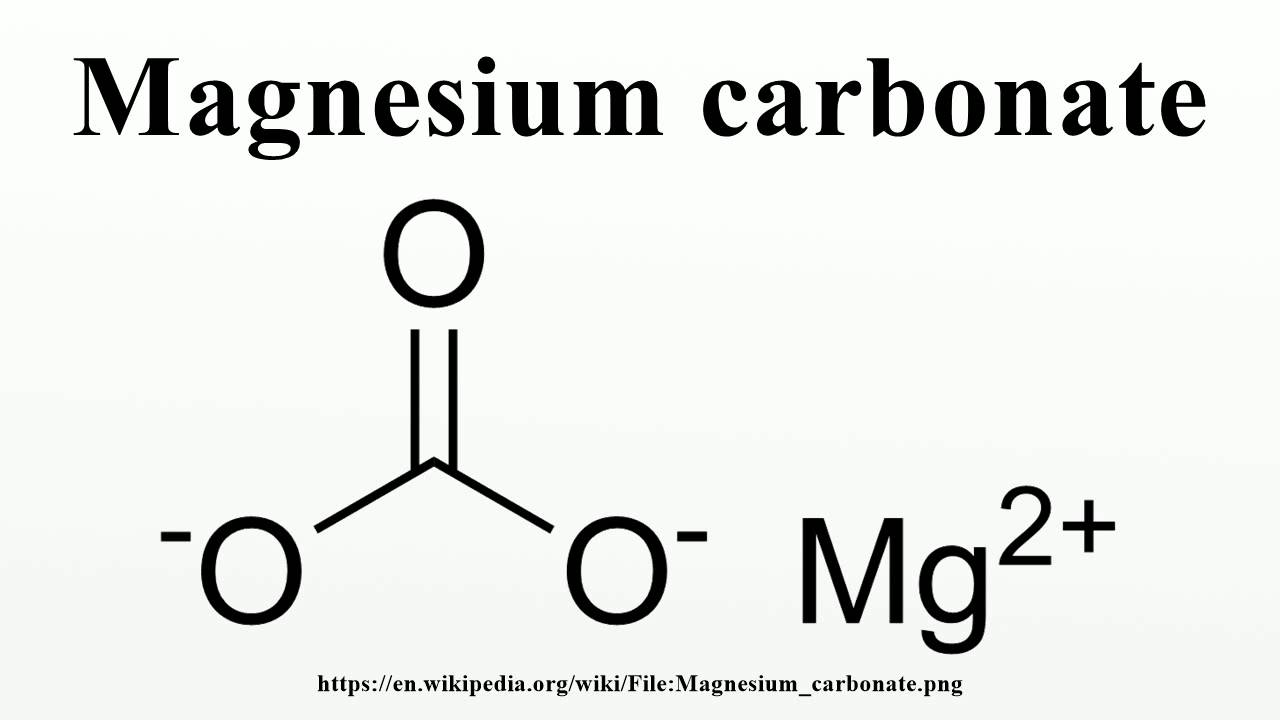
The table below lists the standard Gibbs function of formation for several elements and chemical compounds and is taken from Lange's Handbook of Chemistry. The standard Gibbs free energy of formation ( G f°) of a compound is the change of Gibbs free energy that accompanies the formation of 1 mole of a substance in its standard state from its constituent elements in their standard states (the most stable form of the element at 1 bar of pressure and the specified temperature, usually 298.15 K or 25 ☌). Please help improve this article by introducing citations to additional sources.įind sources: "Standard Gibbs free energy of formation" – news Relevant discussion may be found on the talk page. This article relies largely or entirely on a single source.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
